SCALPORIN™ R1 Blog Series Part 3
A professional scalp device creates the most value when it fits into a broader treatment system.
Technical specifications matter, but they are only one part of the picture. In real treatment environments, professionals evaluate devices based on how well they support consultation, treatment planning, customization, comfort, and workflow efficiency. A device becomes truly meaningful when it works as part of an integrated treatment approach rather than as an isolated tool.
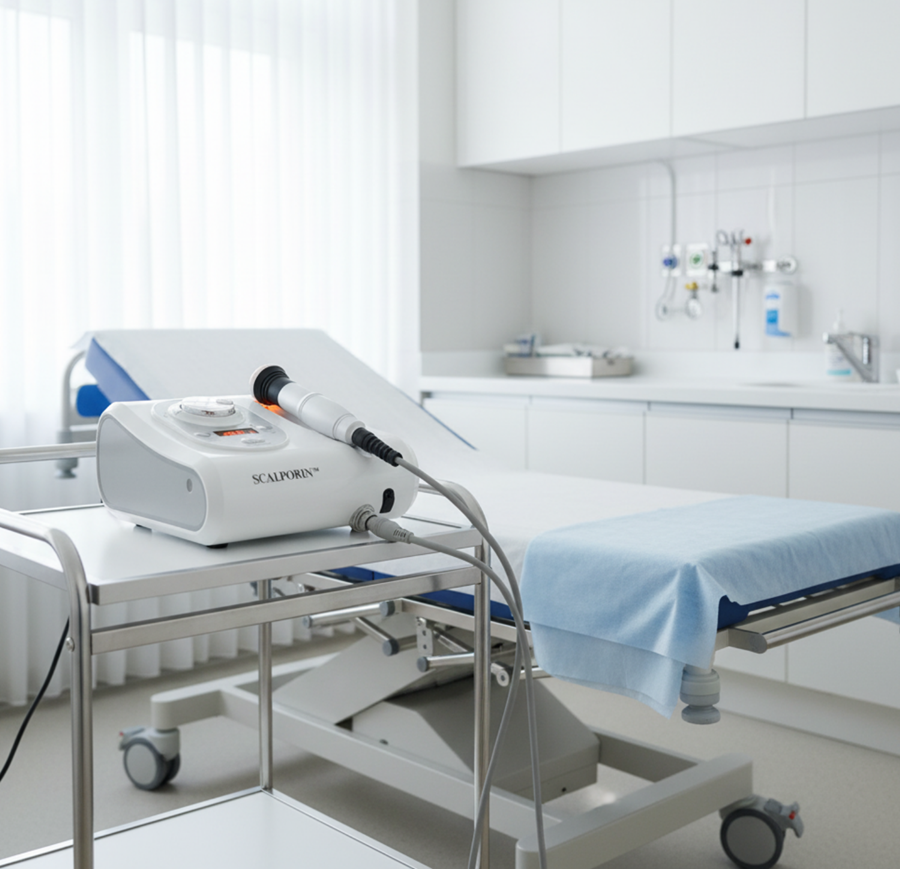
This is the context in which SCALPORIN™ R1 should be understood.
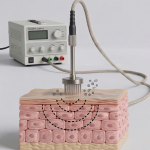
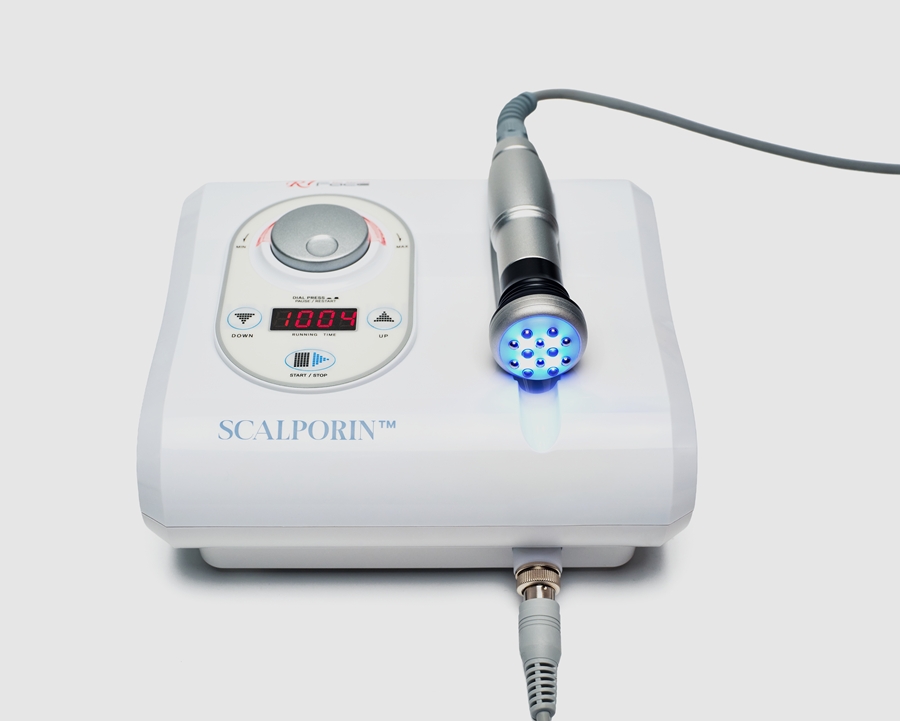
At the functional level, SCALPORIN™ R1 is a professional electroporation scalp device designed to support active ingredient delivery through a non-invasive, needle-free approach. But its professional value extends beyond delivery alone. It is also designed to fit naturally into treatment settings where scalp care is individualized, protocol-based, and expected to deliver both technical credibility and practical usability.
Professional scalp treatment begins with assessment. Practitioners consider scalp condition, treatment goals, the type of active solution being used, and the most suitable treatment structure for the individual case. Because no two clients present exactly the same needs, customization becomes essential.
This is one reason SCALPORIN™ R1 has strategic value. With its wide frequency range and intuitive control, it supports a more tailored treatment experience. It allows practitioners to position scalp care not simply as application, but as a more precise and structured professional procedure.
Comfort also matters in workflow design. A procedure that is needle-free, non-invasive, and associated with no pain and no downtime can be easier to introduce, easier to repeat, and easier to incorporate into ongoing treatment programs. For clinics and professional scalp care centers, this supports both client acceptance and operational consistency.
Another important dimension is protocol fit.
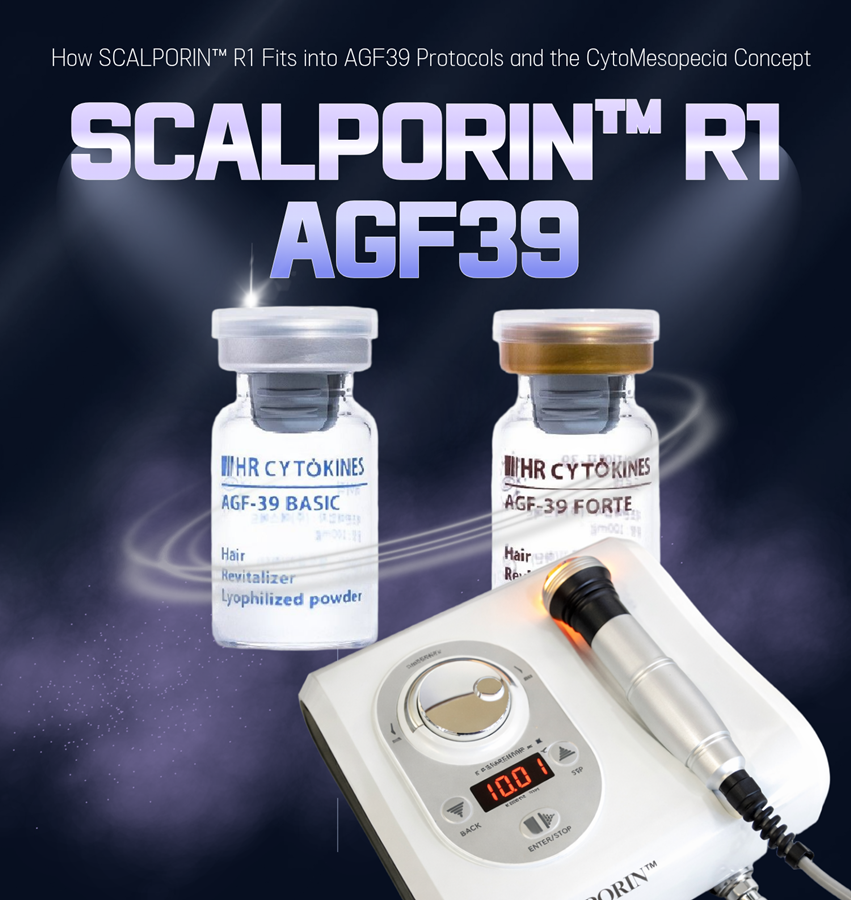
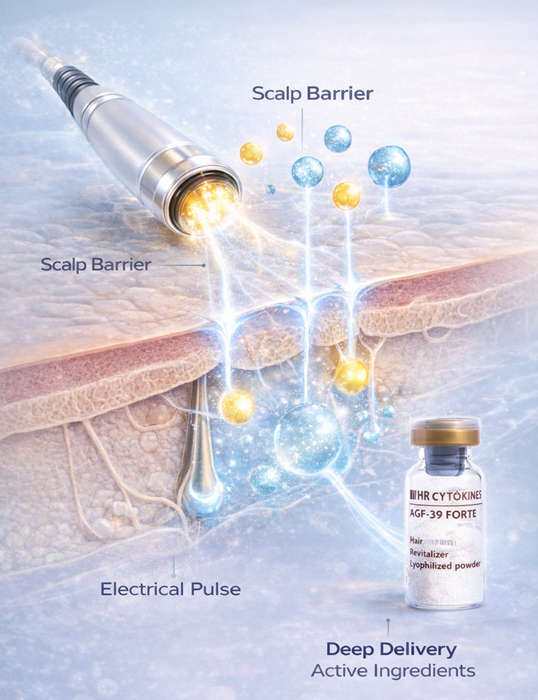
In advanced scalp treatment, a device is rarely used for its own sake. It is used because it supports the treatment logic built around active solutions and clinical objectives. This is where SCALPORIN™ R1 may fit meaningfully within protocols that also involve professional scalp solutions such as AGF39.
Within that relationship, each element has a distinct role.
AGF39 is a professional scalp solution that may be used where a structured scalp treatment protocol is desired.
SCALPORIN™ R1 is the electroporation-based delivery-support device designed to complement such treatment logic.
Together, they may be positioned as part of a more integrated professional scalp care approach.
This broader logic becomes even clearer when viewed through CytoMesopecia – Cytokines Mesotherapy for Alopecia.
CytoMesopecia can be understood as AesMed’s structured professional concept for cytokines-based scalp treatment. It is not simply a product label or a standalone procedural term. Rather, it reflects a treatment framework that combines three essential components:
- active ingredient science,
- delivery logic,
- and workflow-based clinical application.
That framework is important because it reflects how modern professional scalp care is evolving. The strongest treatment systems are not built around isolated features. They are built around coherence. The formulation should make sense. The delivery method should make sense. The treatment sequence should make sense. And the overall professional experience should make sense.


CytoMesopecia represents that type of structured thinking.
Within this framework, SCALPORIN™ R1 can be positioned as the device component that supports the delivery side of cytokines-based scalp treatment logic. AGF39 may be used as part of the solution side of that logic in appropriate professional protocols. CytoMesopecia then provides the conceptual structure that connects these elements into a more complete professional treatment model.
This is why SCALPORIN™ R1 should not be understood only as an electroporation device. It also reflects AesMed’s broader identity as a professional scalp care brand that integrates solutions, devices, and treatment concepts into workflow-based expertise.
For clinics and partners, this distinction matters.
A device alone may be interesting.
A device that fits into a structured professional ecosystem is more valuable.
It is easier to explain.
It is easier to position.
And it is easier to develop into a differentiated treatment offering.
This is the broader value of SCALPORIN™ R1.
It supports delivery.
It supports customization.
It supports workflow.
And within AesMed’s ecosystem, it supports a more integrated professional scalp care philosophy.
The future of professional scalp care will be shaped not by isolated features, but by integrated systems that connect active solutions, delivery technology, and treatment logic. In that sense, SCALPORIN™ R1 is more than a device. It is part of AesMed’s broader vision for structured, precision-based, and professionally differentiated scalp treatment.
To learn more about SCALPORIN™ R1, AGF39, and CytoMesopecia – Cytokines Mesotherapy for Alopecia, explore more expert content on the AesMed official blog or connect with AesMed for professional inquiries and partnership opportunities.