In hair care, trust is built when ideas are tested in a way that others can understand, evaluate, and reproduce. That is why peer-reviewed clinical evidence matters not as a guarantee for every individual, but as a structured signal that a concept was assessed under defined conditions.
In this second post of our series, we walk through a 2012 peer-reviewed pilot study that evaluated the topical application of growth factors followed by microneedle therapy in women with female pattern hair loss (FPHL).
This study is closely connected to the origin story of SGF57, the original blueprint behind AesMed’s evidence-first approach.
Our goal here is simple: explain the study in plain language from design → method → outcomes → safety → meaning so readers can see what was tested and what was observed, without overstatement.
What Was Tested and Why the Design Matters
Why study design is the foundation of credibility
Hair-related outcomes can be influenced by many variables: genetics, baseline severity, stress, seasonality, and measurement inconsistency. A well-chosen study design helps reduce noise and makes the observations easier to interpret.
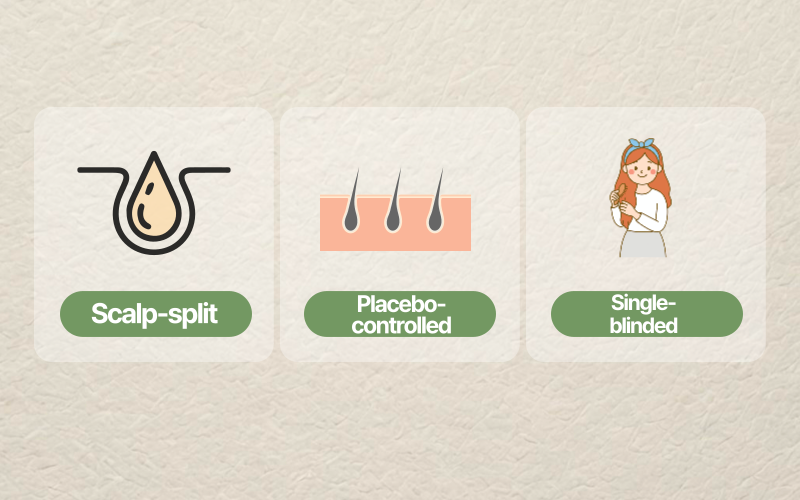
In this study, the authors used a scalp-split, single-blinded, placebo-controlled trial design.
In practice, that means:
- Scalp-split: each participant’s scalp was divided into two halves; one half received the active topical solution and the other half served as a control.
- Placebo-controlled: the control side used normal saline, allowing a comparison against a non-active topical.
- Single-blinded: an investigator who counted hair shafts was blinded to the study condition, helping reduce measurement bias.
This is a practical and meaningful approach for a pilot study because it minimizes differences between individuals. Each person acts as their own reference point, which can be especially helpful when the sample size is limited.
Who participated?
The study enrolled 11 Korean women with Ludwig grade I FPHL (mean age 41.36 ± 2.43 years).
The authors excluded patients who had used medications that could affect the hair cycle within six months, and differential diagnoses were evaluated by a dermatologist.
The protocol was approved by an Institutional Review Board (IRB).
These details matter because they clarify what the study results can reasonably be associated with and what they cannot.
Treatment Protocol: How SGF57 Was Applied
The topical solution (SGF57) and its major components
The treated side received a growth factor solution identified as SGF57, applied topically.
The paper lists major components such as bFGF, IGF-1, VEGF, SCF, KGF-2, SOD-1, and Noggin, along with concentrations.
For readers, the key point is not memorizing the list, but understanding the “why”: the formulation reflects a multi-factor approach aligned with the idea that hair follicle biology is influenced by multiple signaling pathways.
Why microneedling was included
After topical application, both sides of the scalp underwent microneedle therapy.
The authors explicitly state that microneedle therapy was used to enhance drug penetration, noting that microneedles increase skin permeability by creating micro-holes across the stratum corneum.
In other words, the study was not merely asking, “Do growth factors work?” It was testing a combined concept: topical growth factors + a delivery method designed to support penetration.

Device settings and schedule
Each participant received five treatments at weekly intervals.
The microneedling device used nine 33-G microneedles, operating at a 0.5 mm depth and a constant rotational speed of 1500 rpm.
These protocol details are important because clinical credibility is strengthened when methods are clearly described and can be reproduced in future studies.
Outcomes: Hair Density, Satisfaction, and Safety
How outcomes were measured (phototrichogram approach)
To reduce measurement inconsistency, the study used standardized photography conditions and measured a defined scalp location (based on anatomical reference lines).
A digital microscope was used to capture phototrichogram images, and a blinded investigator counted the number of hair shafts using these images.
This matters because it moves beyond subjective impressions and provides a structured method for counting hair shafts in a fixed region.
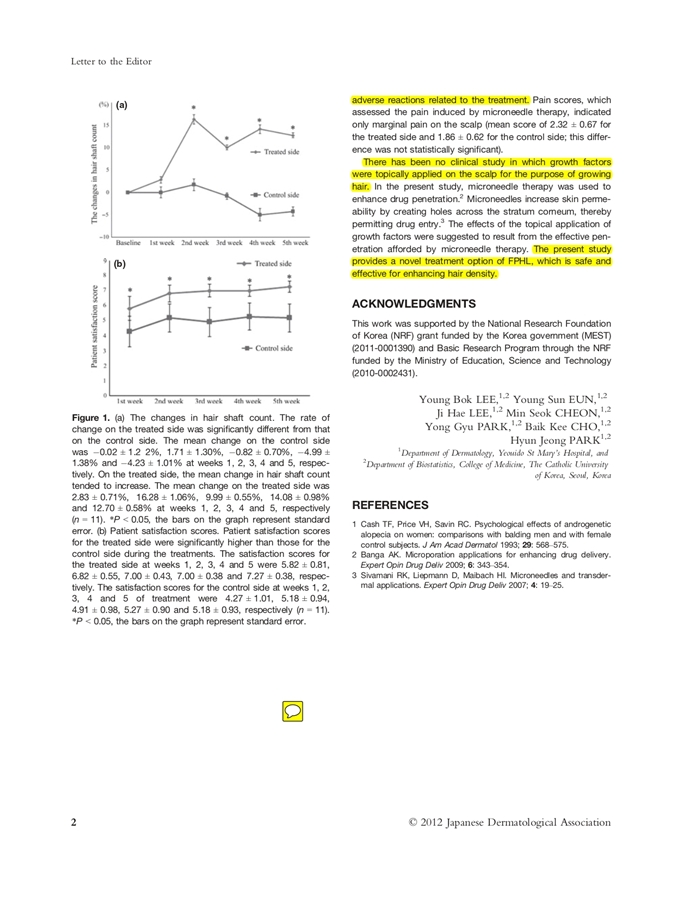
What was observed in hair shaft density
At baseline, mean hair shaft densities were not significantly different between the treated and control halves.
However, differences became significant at specific time points, and the paper reports that:
- An increase of more than 10% compared with baseline was observed on the treated side.
- Mean changes were significantly different between treated and control sides at weeks 2–5.
- Table data show statistically significant differences in mean hair shaft counts at weeks 2, 4, and 5.
The study also provides mean change rates over time, indicating that the treated side tended to increase while the control side showed negative changes at later weeks.
A responsible interpretation is: within this pilot design and short time frame, improved hair shaft count/density was observed on the treated side compared with the control side.
Patient satisfaction
Patients completed satisfaction questionnaires for each side of the scalp.
The mean satisfaction score was higher on the treated side (6.78 ± 0.51) than on the control side (4.96 ± 0.95).
Satisfaction scores for the treated side were reported to be significantly higher during treatments.
Again, satisfaction is not a substitute for objective measurement, but it helps show whether participants perceived a meaningful difference between sides.
Safety and discomfort
A key part of evidence is what did not happen. The paper states there were no adverse reactions related to the treatment.
Pain scores indicated only marginal pain on the scalp, and the difference between treated and control sides was not statistically significant.
This safety note is especially relevant when discussing procedures that involve physical stimulation of the scalp.
What This Means for Evidence-Driven Hair Care (Bridge to AGF39)
What the study suggests without overclaiming
This was a small, short-term pilot study. Yet it provides several meaningful signals:
- The concept of topical growth factors + microneedling delivery was tested under a controlled design.
- The authors suggest that the effects of growth factors may be linked to effective penetration afforded by microneedle therapy.
- The study describes the approach as a novel option that was safe and effective for enhancing hair density (in the context of the pilot).
The right takeaway is not “guaranteed regrowth.” The right takeaway is that the study provides structured evidence: clear protocol, objective measurement, a control condition, and safety reporting.
How this connects to AGF39 responsibly
SGF57 is widely referenced as the original blueprint behind later evidence-driven approaches. In AesMed’s language, that matters because credibility is cumulative: a company’s later frameworks are more trustworthy when they are built on a history of transparent testing and measurement.
In that sense, AGF39 is built on the same evidence-first philosophy a commitment to defined protocols, measurable outcomes, and academic communication rather than vague claims. (This post does not claim direct equivalence of products or identical clinical outcomes; it highlights the lineage of evidence-focused thinking.)

Limitations & Why Follow-up Evidence Matters
Because the study enrolled only 11 participants and followed them across five weekly sessions, it should be interpreted as an early signal not a final answer.
Larger studies, longer follow-up periods, and expanded participant groups can help clarify:
- durability of observed changes
- optimal treatment intervals
- how outcomes vary across severity levels
- and which protocol parameters matter most
That is exactly why subsequent conference presentations (2013 and 2014) explored broader clinical experience and parameters such as microneedle depth.
2014 WCHR poster.

“Some of the images were created using Miricanvas and Gemini.”










